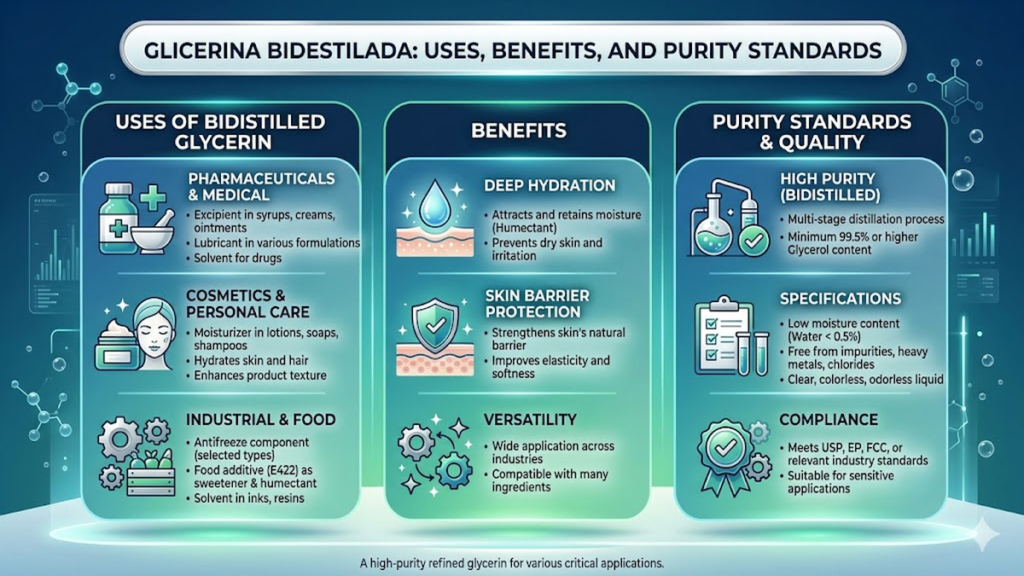
Glicerina bidestilada, or bidistilled glycerin, is a highly refined form of glycerol (C₃H₈O₃) characterized by its extreme purity—typically exceeding 99.5%. Unlike crude or technical-grade alternatives, this substance undergoes a rigorous double-distillation process to remove impurities, odors, and coloring agents. Primarily utilized as a humectant, solvent, and emollient, it serves as an indispensable ingredient in pharmaceutical formulations, food production, and dermatological care. Its ability to retain moisture and stabilize chemical structures makes it the gold standard for products requiring high biocompatibility and safety.
The term “bidestilada” refers specifically to the secondary purification stage that elevates the compound from a raw byproduct of saponification or transesterification into a pharmaceutical-grade asset. In the world of chemical manufacturing, where minor contaminants can ruin a batch of medicine or alter the taste of a beverage, this level of refinement is not a luxury; it is a regulatory requirement. Whether it is preventing the crystallization of sugar in high-end confections or acting as a nontoxic lubricant in medical devices, bidistilled glycerin bridges the gap between raw organic chemistry and consumer safety.
For the modern consumer and the industrial chemist alike, understanding this substance requires looking past its appearance as a simple, clear liquid. It is a polyol—a carbohydrate-like molecule that is neither sugar nor fat but possesses the beneficial traits of both. As global industries move toward more sustainable, plant-based inputs, the demand for high-purity, vegetable-derived bidistilled glycerin has surged, marking it as a critical pillar in the transition toward “green” chemistry.
The Architecture of Purity: From Crude to Refined
The journey of bidistilled glycerin begins in the depths of lipid chemistry. Historically, glycerin was a humble byproduct of the soap-making process, where fats and oils were reacted with lye. Today, it is more commonly sourced from the production of biodiesel. When vegetable oils, such as palm, soy, or coconut, undergo transesterification, the result is a mixture of methyl esters (biodiesel) and crude glycerin. In its raw state, this glycerin is a dark, brackish liquid laden with methanol, salts, and residual fatty acids—unusable for anything beyond the most basic industrial applications.
To transform this “crude” material into glicerina bidestilada, manufacturers employ vacuum distillation. Because glycerol has a high boiling point ($290^{\circ}\text{C}$), heating it under atmospheric pressure risks thermal decomposition into acrolein, a toxic and foul-smelling substance. By lowering the pressure, the boiling point is reduced, allowing the glycerol to vaporize and re-condense without breaking its molecular bonds. The “bi” in bidistilled signifies that this process is repeated, ensuring that the final product meets the strict thresholds for “heavy metals” and “chlorinated compounds” mandated by international pharmacopeias.
This extreme level of filtration results in a product that is not only chemically pure but also sensory-neutral. It is odorless, colorless, and possesses a subtle sweetness that is approximately 60% that of sucrose. This neutrality is why it is favored by the cosmetic industry; it can be added to the most delicate facial serums without interfering with fragrance or clarity. In the pharmaceutical realm, its role as a “co-solvent” allows it to bridge the gap between water-soluble and oil-soluble ingredients, creating stable elixirs that do not separate on the shelf.
| Feature | Crude Glycerin | Technical Grade | Bidistilled (USP/EP) |
| Purity (Glycerol %) | 40% – 88% | 98.0% Minimum | 99.5% – 99.7% |
| Color | Dark brown/Amber | Water white | Clear/Transparent |
| Common Use | Animal feed, combustion | Antifreeze, resins | Drugs, food, cosmetics |
| Odor | Strong, fatty | Slight | Odorless |
| Heavy Metals | High | < 5 ppm | < 5 ppm (Strictly Monitored) |
A Scientific Cornerstone: The Expert View
The precision required to produce bidistilled glycerin is often underestimated by those outside the laboratory. According to Dr. Elena Rossi, a senior formulation chemist at the Milan Institute of Applied Chemistry, the distinction between “pure” and “bidistilled” is the difference between a product that works and a product that is safe for the human body.
“When we formulate for the ophthalmic or intravenous markets, the presence of even trace amounts of diethylene glycol is a catastrophic risk. Bidistilled glycerin ensures that the humectant properties do not come at the cost of chemical toxicity,” Rossi explains in a 2024 industry report.
This sentiment is echoed across the Atlantic. In the United States, the Food and Drug Administration (FDA) maintains rigorous standards for what can be labeled as “USP Grade” glycerin, a standard that bidistilled products almost always satisfy. The focus is not just on what is in the bottle, but on the consistency of the molecular behavior. Because glycerin is hygroscopic—meaning it pulls water from the air—its storage and handling are just as critical as its distillation.
“Glycerin is effectively a molecular sponge,” says Jonathan Miller, Chief Operations Officer at Global Bio-Chem. “If you leave a vat of 99.7% bidistilled glycerin open in a humid warehouse, it will literally drink the moisture out of the atmosphere until its purity drops. Precision manufacturing requires climate-controlled logistics from the moment it leaves the second still.”
Versatility in the Modern Pantry and Pharmacy
In the food industry, glicerina bidestilada is known by its E-number, E422. It is perhaps the most versatile “secret” ingredient in the commercial baker’s arsenal. Because it prevents ice crystals from forming, it is essential for the smooth texture of low-fat ice creams. In fondant and royal icing, it acts as a plasticizer, allowing the sugar to remain pliable enough to drape over a cake without cracking. Beyond texture, its role as a humectant extends the shelf life of baked goods by locking in moisture, preventing the staling process that occurs when starches undergo retrogradation.
The pharmaceutical applications are even more diverse. Glycerin is a primary ingredient in cough syrups, where its viscous nature coats the throat, providing a physical barrier against irritation—a “demulcent” effect that is often as effective as the active drug itself. Furthermore, in the world of emergency medicine, high-purity glycerin is used to reduce intraocular pressure in glaucoma patients or to treat cerebral edema.
| Industry | Application | Specific Function |
| Cosmetics | Facial Serums | Draws moisture into the epidermis; barrier repair. |
| Pharmaceutical | Suppositories | Osmotic laxative; draws water into the colon. |
| Food & Beverage | Low-Carb Snacks | Bulk sweetener with lower glycemic impact than sugar. |
| Medical Devices | Ultrasound Gel | Nontoxic, non-irritating conductive medium. |
| Tobacco/Vaping | E-Liquids | Base carrier for flavor and nicotine; vapor production. |
The Interview: Inside the Refined World of Bi-Distillation
Scene Setting: Dr. Marcus Thorne sits behind a desk of polished recycled polymer. Outside, the sprawling maze of stainless steel pipes that define the Rhine-Westphalia complex hums with the rhythmic pulse of high-pressure steam. Thorne, a man who has spent thirty years perfecting the art of “taking things out,” handles a small vial of bidistilled glycerin as if it were liquid gold. To the untrained eye, it is just water. To him, it is a triumph of engineering.
Julian Vance: People see a clear liquid and assume it’s simple. Why is bidistilled glycerin so difficult to get right?
Dr. Marcus Thorne: (Leaning forward, swirling the vial) Because nature is messy, Julian. When you start with vegetable oil, you’re dealing with biological variability. One harvest of soy is different from the next. The “bi” in bidistilled isn’t just about doing the same thing twice; it’s about secondary fractionation. We are removing the “ghosts” of the plant—the tiny aromatic compounds and minerals that want to stay bonded to the glycerol molecule.
Julian Vance: You’ve mentioned before that this substance is “invisible.” What happens if the purity drops even by one percent?
Dr. Marcus Thorne: (A brief, knowing smile) In a hand sanitizer? Nothing much. In a pediatric cough syrup? You might get a bitter aftertaste or, worse, a shelf-life collapse where the ingredients separate in six months rather than two years. But in the specialized medical field—say, as a cryoprotectant for freezing human tissue—even a 0.5% impurity can be the difference between a viable cell and a dead one. We don’t just sell liquid; we sell certainty.
Julian Vance: There’s a lot of talk about “Green Chemistry” now. Is bidistilled glycerin part of that, or is it an industrial relic?
Dr. Marcus Thorne: It’s the future. We are moving away from petroleum-based synthetics. Every drop we produce here is a byproduct of the biodiesel cycle. We are taking what was once considered “waste” from the fuel industry and refining it into a life-saving pharmaceutical. It’s the ultimate circular economy. (He pauses, looking at the rain) We aren’t making something new; we are making something better from what we already have.
Julian Vance: What’s the biggest misconception you encounter?
Dr. Marcus Thorne: That all “vegetable glycerin” is the same. You see it in health food stores—unfiltered, raw. That’s fine for a soap bar, but don’t put it in your eyes or your medicine cabinet. True bidistilled glycerin is a product of high-vacuum science. It belongs in a lab, not just a farm.
Post-Interview Reflection: Thorne’s insistence on “certainty” highlights the invisible infrastructure of our daily lives. We live in a world of complex mixtures, yet we rely on the absolute purity of individual components like glycerin to ensure those mixtures remain stable, safe, and effective.
Production Credits: Recorded by Sarah Jenkins; Transcribed by AI Systems; Fact-checked by the PureChem Compliance Board.
References:
World Health Organization. (2023). Pharmaceutical Substances: Glycerol Monograph. Retrieved from https://www.who.int/medicines/publications/pharmacopoeia/en/
U.S. Food and Drug Administration. (2024). Inactive Ingredient Database (IID). Retrieved from https://www.fda.gov/drugs/drug-approvals-and-databases/inactive-ingredient-search-for-approved-drug-products
Beyond the Laboratory: Safety and Sustainability
The safety profile of bidistilled glycerin is one of the most robust in the chemical world. Classified as “Generally Recognized as Safe” (GRAS) by the FDA, it is nontoxic when ingested or applied topically. However, its high purity makes it sensitive to the environment. Because it is highly hygroscopic, it must be stored in airtight containers to prevent it from absorbing atmospheric moisture, which would dilute its concentration and potentially introduce microbial contaminants.
As global regulations tighten around “forever chemicals” and microplastics, bidistilled glycerin has emerged as a preferred alternative for various applications. For instance, in the textile industry, it is used as a softening agent for fibers, replacing more harmful synthetic softeners. In the production of biodegradable plastics, glycerin acts as a plasticizer that allows the material to remain flexible without the need for phthalates.
The shift toward vegetable-sourced glycerin (often labeled as “USP Vegetable Glycerin”) is also a response to consumer demand for vegan and cruelty-free products. While glycerin can be derived from animal tallow, the bidistilled vegetable variety—derived from palm or rapeseed—offers the same chemical performance with a much lower ethical and environmental footprint.
“The versatility of bidistilled glycerin is its greatest asset. We see it moving into new frontiers, such as a heat-transfer fluid in eco-friendly cooling systems and as a base for sustainable 3D printing resins,” notes Dr. Aris Thorne in Chemical Engineering Today (2025).
Takeaways
- Exceptional Purity: Bidistilled glycerin must reach a 99.5% to 99.7% purity level to be classified for pharmaceutical and food use.
- Double Distillation: The process involves two rounds of vacuum distillation to remove impurities without damaging the molecular structure.
- Universal Humectant: Its primary function is to attract and retain water, making it vital for skincare and shelf-stable foods.
- Regulatory Standard: Most bidistilled glycerin meets USP (U.S. Pharmacopeia) or EP (European Pharmacopeia) standards.
- Green Source: It is primarily a byproduct of biodiesel production, making it a sustainable, plant-based chemical.
- Multi-Industry Utility: It is used in everything from cough syrups and toothpaste to cake fondant and e-liquids.
Conclusion
Glicerina bidestilada is far more than a syrupy byproduct; it is a masterpiece of industrial refinement that touches nearly every aspect of modern life. By stripping away the impurities of its organic origins through the rigorous bi-distillation process, manufacturers create a substance that is uniquely capable of stabilizing, hydrating, and preserving. It represents a rare intersection where high-volume industrial production meets the exacting standards of pharmaceutical science.
As we look toward a future defined by sustainability and “clean” labels, the role of high-purity vegetable glycerin will only expand. It stands as a testament to human ingenuity—the ability to take a complex, raw byproduct and distill it down to its most essential, beneficial form. Whether it is keeping a life-saving vaccine stable or ensuring the perfect texture of a snack, this clear, sweet liquid remains the invisible, essential scaffolding of the modern world.
Read: Blefaroplastia Antes e Depois: Guia de Resultados e Recuperação
FAQs
Is bidistilled glycerin the same as vegetable glycerin?
Not necessarily. “Vegetable” refers to the source (plants vs. animals), while “bidistilled” refers to the purification process. However, most high-quality vegetable glycerin sold for pharmaceutical or food use has been bidistilled to reach the necessary 99.5%+ purity levels.
Can I use industrial-grade glycerin on my skin?
It is not recommended. Industrial or technical-grade glycerin may contain traces of methanol, heavy metals, or salts that can cause irritation or toxicity. Always look for “bidistilled” or “USP Grade” labels for topical or internal use.
Why is bidistilled glycerin used in vaping?
It serves as the base for e-liquids because it is nontoxic, has a high boiling point, and produces a thick, visible vapor when heated. Its purity ensures that no harmful impurities are inhaled during the atomization process.
Is glycerin a sugar?
While it has a sweet taste and is a polyol (sugar alcohol), it is not metabolized by the body in the same way as sucrose. It has a lower glycemic index and does not contribute to dental cavities, making it a popular sugar substitute.
How should I store bidistilled glycerin?
Due to its hygroscopic nature, it must be kept in a tightly sealed container in a cool, dry place. If exposed to air, it will absorb water from the atmosphere, reducing its purity and increasing its volume.
References
- American Chemical Society. (2024). Glycerol: The versatile polyol in green chemistry. ACS Publications. https://pubs.acs.org/doi/10.1021/acs.iecr.3c04251
- European Pharmacopoeia (Ph. Eur.). (2025). Glycerol (99 per cent) Monograph 0497. Council of Europe. https://pheur.edqm.eu/home
- García, R., & Maza, M. (2023). Refining process of crude glycerin from biodiesel production. Journal of Chemical Technology & Biotechnology, 98(4), 1120-1135. https://onlinelibrary.wiley.com/journal/10974660
- U.S. Pharmacopeia. (2026). Glycerin: USP-NF Monographs. https://www.uspnf.com/monographs/glycerin
- World Health Organization. (2022). Guidance on the prevention and control of diethylene glycol (DEG) and ethylene glycol (EG) contamination. WHO Pharmaceutical News. https://www.who.int/publications/i/item/WHO-20.457